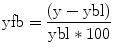Study
Participants
Age (years)
BMI (kg/m2)
Duration of illness/recovery
Medication
Additional relevant information (e.g., specific exclusion criteria, report on ED severity measure)
Boghi et al. (2011)
AN-R: n = 21
AN-R: 29 ± 10.1
AN-R: 15.5 ± 1.8
Illness (in years):
All patients on SSRI treatment
Exclusion criterion: past or current alcohol or substance abuse
“Short” duration: n = 10
HC: 30.8 ± 8.7
HC: 21.9 ± 1.5
AN-R: 11.3 ± 12.1
No measure of ED severity reported
“Long” duration: n = 11
“Short”: 1.9 ± 1.3
HC: n = 27 (Age-matched)
“Long”: 19.8 ± 11.1
Brooks et al. (2011a)
AN: n = 14
AN: 26 ± 7.1
AN: 15.6 ± 1.5
Illness (in years):
NR
ED severity measure: Eating Disorder Examination Questionnaire
“AN-R: n = 8”
“AN-R: 26 ± 10.9”
“AN-R: 15.1 ± 1.9”
AN: 9.2 ± 7.1
“AN-BP: n = 6”
“AN-BP: 27 ± 9.7”
“AN-BP: 16.2 ± 1.1”
“AN-R: 9.2 ± 11.6”
HC: n = 21 (Age-matched)
HC: 26 ± 9.6
HC: 21.4 ± 2.3
“AN-BP: 9.2 ± 8.2”
Castro Fornieles et al. (2009)
AN: n = 12
AN: 14.5 ± 1.5
AN (baseline): 14.8 ± 2.0
NR
1 at baseline and 3 at follow-up on fluoxetine or fluvoxamine
One male in each group
(9 AN-R; 3 AN-BP)
HC: 14.6 ± 3.2
AN (follow-up): 18.8 ± 0.4
ED severity measure: Eating Attitudes Test
HC: n = 9
HC: not reported
Gaudio et al. (2011)
AN-R: n = 16
AN-R: 15.2 ± 1.7
AN-R: 14.2 ± 1.4
Illness (in months):
All on clomipramine or SSRI (duration of treatment: 16.4 ± 6.4 days)
Exclusion criterion: current or past other DSM-IV-TR disorders
HC: n = 16
HC: 15.1 ± 1.5
HC: 20.2 ± 1.6
AN-R 5.3 ± 3.2
10 on haloperidol (0.5–2.0 mg/day; mean dose: 1.3 ± 0.5 mg/day; mean duration of treatment: 9.2 ± 6.4 days)
ED severity measure: Eating Attitudes Test
Joos et al. (2010)
AN-R: n = 12
AN-R: 25.0 ± 4.8
AN-R: 16.0 ± 1.2
Illness (in years):
AN-R:1 on sertraline 75 mg/day
AN-R group: 1 participant had “inconstant bulimic phases” and 1 used laxatives
BN: n = 17
BN: 24.5 ± 4.8
BN: 21.1 ± 2.5
AN-R: 4.7 ± 3.6 years
ED severity measure: Eating Disorder Inventory
HC: n = 18 (Age-matched)
HC: 26.9 ± 5.7
HC: 21.2 ± 2.0
BN: 7.5 ± 5.7 years
Muhlau et al. (2007)
rAN-R: n = 22
rAN-R: median 22.3 (range: 18.4–40.8)
rAN-R: median 19.5 (range: 17.0–22.8)
Recovery (in months):
NR
Exclusion criteria: lifetime history of post-traumatic stress disorder, manic episodes, schizophrenia, obsessive compulsive disorder, substance use disorders, or borderline personality disorder. Major depressive disorder occurring outside episodes of low weight was also an exclusion criterion. During episodes of low weight at least 16 rAN-R, participants had met the criteria for depressive disorder at least once
HC: n = 37
HC: median 23.8 (range: 18.3–40.2)
HC: 20.1 median 22.3 (range: 18.3–24.8)
rAN-R: median 15.5 (range: 6–60)
Lifetime lowest BMI: median 13.5 (range: 10.0–16.1)
Duration of AN (years): median 5 (range: 1–23)
Roberto et al. (2011)
AN: n = 32
AN: 26.9 ± 6.4
AN (baseline): 16.0 ± 1.6
Illness (in years):
No medication
Exclusion criteria: “axis I disorder other than major depression” and “history of suicide attempt or other self-injurious behavior within the previous 6 months”
(14 AN-R; 18 AN-BP)
HC: 25.0 ± 3.2
AN (follow-up): 20.0 ± 0.6
AN: 10.2 ± 6.2
Amenorrhea was not considered as a criterion for AN
HC: n = 21
HC (baseline): 20.8 ± 1.2
HC (follow-up): 20.6 ± 1.2
Schafer et al. (2010)
BN-P: n = 14
BN: 23.1 ± 3.8
BN: 22.1 ± 2.5
Illness (in years):
No medication
Exclusion criterion: “clinically relevant depression”
BED: n = 17
BED: 26.4 ± 6.4
BED: 32.2 ± 4.0
BN: 7.3 ± 3.6
ED severity measure: Eating Disorder Inventory
HC: n = 19
HC: 22.3 ± 2.6
HC: 21.7 ± 1.4
BED: 6.8 ± 4.0
Suchan et al. (2010)
AN: n = 15
AN: 26.8 ± 8.4
AN: 16.0 ± 1.3
Illness (in years):
NR
No measure of ED severity reported
HC: n = 15
HC: 29.5 ± 8.2
HC: 22.0 ± 2.1
AN: 5.5 ± 5
Wagner et al. (2006)
rAN-R: n = 14
rANR: 23.7 ± 5.3
rANR: 21.2 ± 2.0
Recovery (in months):
No medication
Comorbid axis I and II assessed but not included in report
rAN-BP: n = 16
rANBP: 27.4 ± 7.2
rANBP: 21.2 ± 1.5
rAN-R: 28.7 ± 20.4
Lifetime lowest BMI:
rBN: n = 10
rBN: 24.0 ± 6.1
rBN: 23.1 ± 2.4
rAN-BP: 39.5 ± 52.7
rANR: 14.1 ± 1.4
HC: n = 31
HC: 26.8 ± 7.3
HC: 21.9 ± 2.0
rBN: 29.8 ± 18.1
rANBP: 14.8 ± 2.0
rBN: 19.2 ± 2.1
HC: 20.1 ± 1.4
18.3 Functional Studies
In this section, we review functional MRI studies according to the different aspects of the eating disorders psychopathology investigated. Eating disorders involve changes in eating behavior and usually, but not always, are associated with overvalued ideas about shape and weight. Additional features include abnormalities in cognitive style, emotional regulation, social functioning, reward sensitivity, and interoceptive awareness (Treasure et al. 2011).
18.3.1 Eating
Abnormal eating behaviors lie at the heart of all forms of EDs. This includes both over and under control of eating, sometimes associated with the interruption of the process of digestion by spitting/vomiting. Table 18.2 summarizes neuroimaging findings on functional activations to food stimuli in people with EDs.
Table 18.2
Summary of neuroimaging findings on functional activations to food stimuli in people with eating disorders
Study | Task | Sample | Main findings | Interpretation |
|---|---|---|---|---|
Bohon and Stice (2011) | Gustatory paradigm (palatable vs. neutral food) | BN vs. subthreshold BN vs. HCs | BN < HCs: right prefrontal gyrus (anticipatory and consummatory), left middle frontal gyrus, left thalamus, right posterior insula (consummatory) | People with bulimic symptoms may experience less activation in gustatory and reward regions during anticipation and receipt of palatable foods |
Brooks et al. (2011b) | Food vs. nonfood pictures | AN vs. BN vs. HCs | HC > AN, BN: right insular cortex, right superior temporal gyrus, left side cerebellum, left caudate body | AN and BN: activation of cognitive control |
AN > BN, HC: left visual cortex, cerebellum, right DLPFC, right precuneus | AN and BN: sustained visual attention to food stimuli (possible substrate for attentional bias) | |||
BN > AN, HC: right visual cortex, right insula, left prefrontal gyrus | BN: greater reward sensitivity | |||
BN < HC: bilateral superior temporal gyrus, insular cortex, left visual cortex | ||||
BN < AN: right parietal lobe, left dorsal posterior cingulate | ||||
BN > AN: right caudate, right superior temporal gyrus, left supplementary motor area | ||||
Burger and Stice (2011) | Gustatory paradigm (palatable food) and food pictures | HCs: high vs. low dietary restraint | High dietary restraint > low dietary restraint (food receipt): OFC, DLPFC | Individuals who report high dietary restraint have a hyperresponsivity in reward-related brain regions when food intake is occurring |
Frank et al. (2011) | Reward learning paradigm (association learning between conditioned visual and unconditioned taste stimuli) | BN vs. HCs | BN < HCs: insula, ventral putamen, amygdala, OFC | Altered temporal learning in BN, which could be due to episodic excessive food stimulation which results in desensitization of dopamine circuits |
Gizewski et al. (2010) | Food vs. nonfood pictures | AN-R vs. HCs | AN = HCs (hunger): insula (anterior insula in AN; posterior insula in BN) | Food stimuli more emotionally arousing to AN, but more physically stimulating to HCs |
Food vs. nonfood pictures | AN-R vs. HCs | AN R > HCs: right amygdala | AN: negative feedback loop of emotional processing (dysfunction of the top-down processes of the dorsal stream) | |
AN R < HCs: cingulate cortex | ||||
Pietrini et al. (2011) | Review | AN-R vs. AN-b/p | AN-R: rest < symptom provocation: frontal cortex; cingulate cortex | Possible disturbance of a network involving frontal, parietal, and cingulate metabolism at rest in AN that normalize after recovery |
AN-B/P: rest < symptom provocation: frontal, parietal, and cingulate | ||||
Santel et al. (2006) | Food vs. nonfood pictures | AN vs. HCs | AN < HCs (satiety): left inferior parietal cortex | Decreased food-related somatosensory processing in AN during satiety. Attentional mechanisms during hunger might facilitate restricted eating |
AN < HCs (hunger): right visual occipital cortex | ||||
Schienle et al. (2009) | Food vs. nonfood pictures | BED vs. BN vs. HCs vs. overweight | Food > nonfood: OFC, ACC, insula | Differential brain activations in reward circuitry associated with food in patients suffering from BED and BN |
BED > overweight, HCs, BN: medial OFC | ||||
BN > overweight, HCs, BED: ACC, insula | ||||
Stice et al. (2010) | Palatable food vs. unpalatable food vs. neutral food pictures | HCs (from lean to obese) | Palatable food < unpalatable/neutral food: frontal operculum, lateral OFC, striatum predicted > BMI for those with DRD2 TaqIA A1 allele or DRD4-7R allele | Responsivity of reward circuitry to food increases risk for future weight gain, with the moderating effect of genes that impact dopamine signaling capacity |
Stice et al. (2011) | Gustatory paradigm (receipt and anticipated receipt of palatable vs. neutral food stimuli) | HCs: high vs. low risk for obesity | High > low-risk obesity (palatable food receipt): caudate, parietal operculum, frontal operculum | Youth at risk for obesity show elevated reward circuitry responsivity in general, coupled with elevated somatosensory region responsivity to food |
No differences in response to anticipated food reward | ||||
Uher et al. (2003) | Food vs. nonfood pictures | Recovered AN-R vs. AN-R vs. HCs | REC > HCs (food): medial prefrontal cortex, dorsal anterior cingulate, cerebellum | Frontal lobe reactivity could be evaluated as a candidate factor predictive of outcome in AN |
REC < HCs (food): left parietal lobule and visual occipital cortex | The brain response in people recovered from AN is a combination of the responses seen in ill patients (medial frontal) and those in HCs (apical and lateral prefrontal) | |||
REC, HCs > AN (food): right lateral PFC, apical PFC, dorsal ACC | ||||
AN > REC, HCs (food): superior medial PFC | ||||
REC, HCs > AN: apical PFC, bilateral DLPFC, medial paracentral cortex | ||||
AN, REC > HCs: medial PFC, cerebellum | ||||
Uher et al. (2004) | Food vs. nonfood pictures | AN vs. BN vs. HCs | HCs > EDs (food): left lateral PFC, left parietal cortex, bilateral visual cortex, cerebellum | Abnormal focus of food-related activity in the medial prefrontal region identified in a large number of ED patients |
EDs > HCs (food): left medial orbitofrontal and anterior cingulate cortices | ||||
ED < HCs (food): lateral PFC, inferior parietal lobule, cerebellum | ||||
BN < HCs (food): lateral and apical PFC | ||||
van Kuyck et al. (2009) | Review | AN-R | Parietal cortex: < | No specific imaging biomarker or pattern for AN discovered |
Anterior and subgenual cingulate cortex: < rest; > symptom provocation | ||||
Temporal lobe: symptom provocation: > insula; amygdala | ||||
Vocks et al. (2011) | Gustatory paradigm (chocolate vs. hunger vs. satiety) | AN vs. HCs | AN > HCs (chocolate + hunger): amygdala and left medial temporal gyrus | AN: fear response to high-calorie food |
HC > AN (chocolate + hunger): right medial frontal gyrus | HC: high reward anticipation | |||
Wagner et al. (2008) | Gustatory paradigm (sugar vs. water) | Recovered AN-R vs. HCs | Recovered AN < HCs: insula, dorsal and middle caudate, dorsal and ventral putamen, anterior cingulate | Individuals recovered from anorexia have disturbances of gustatory processing |
AN = HCs: anterioventral striatum, amygdala, and OFC |
18.3.1.1 Eating in Anorexia Nervosa
A recent review of neuroimaging studies involving food-related stimuli in EDs concludes that there are altered activations in the parietal and temporal cortices, anterior and subgenual cingulate cortex, and frontal cortex in AN (van Kuyck et al. 2009). A meta-analysis of functional MRI studies using coordinate-based meta-analysis methodology reports increased activation of medial frontal and caudate regions and reduced activation in parietal areas in AN during exposure to food cues (Zhu et al. 2012). In restricting AN, food stimuli are associated with the activity in the brain network responsible for the identification of emotional significance of the stimuli, affective states, and autonomic regulation (e.g., bottom-up processes), including the right amygdala (Joos et al. 2011b), ventral striatum (Wagner et al. 2008), orbitofrontal cortex (Uher et al. 2004), and insular cortex (Uher et al. 2004; Gizewski et al. 2010; Schienle et al. 2009; Vocks et al. 2010). Also, food stimuli are associated with abnormalities in brain areas involved in regulatory processes (e.g., top-down), comprising dorsal regions of the anterior cingulate cortex, posterior cingulate cortex (Gizewski et al. 2010), prefrontal cortex (Pietrini et al. 2011), medial prefrontal cortex (Uher et al. 2004), and dorsolateral prefrontal cortex (Brooks et al. 2011b




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree