Fig. 1
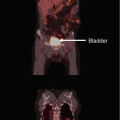
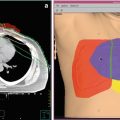
(a) A 75-year-old Caucasian male who previously received 50 Gy in 20 daily fractions using superficial energy photons in the setting of a squamous cell carcinoma. Note the infield hypopigmentation and epidermal atrophy delineating his radiotherapy field. Anterior to this the patient now has a biopsy-proven new cutaneous squamous cell carcinoma which encroaches over the previous field. The need to overlap any further radiotherapy field meant the patient was recommended surgery. (b) The patient underwent wide excision and free tissue graft reconstruction. Despite this the pathology was unfavourable with a close deep excision margin and the presence of perineural invasion. The patient was recommended adjuvant radiotherapy to a dose of 50 Gy in 20 daily fractions using orthovoltage energy photons. Because of the removal of the previously irradiated tissue, the risk of re-irradiation was deemed negligible
4.2 Infield Recurrence After Radiotherapy for Non-melanoma Skin Cancer
Infield recurrence after RT for NMSC is not common. A typical scenario for infield recurrence when it does occur is local recurrence of a midface BCC after previous definitive RT. A clinical scenario such as this, particularly in the setting of a deep recurrence located in the midface, can pose a difficult management problem (Smith and Grande 1991), and patients may be better approached with salvage surgery rather than further RT.
Presentations of new NMSC adjacent to a previously irradiated site are a more common scenario as are recurrences at the edge of a previously treated RT field (marginal recurrence). These patients may be candidates for further RT, and ideally the details of previous treatment including the prescribed dose/fractionation schedule should be obtained, but are not always available.
Clinical assessment is very important, and patients exhibiting obvious late RT changes such as infield cutaneous hypo- (or hyper)pigmentation, telangiectasia, epidermal atrophy or fibrosis (Fig. 2) are not optimal candidates for re-irradiation.
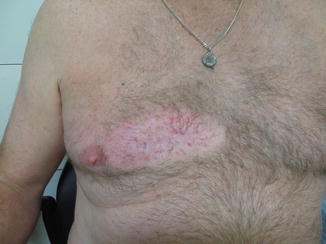

Fig. 2
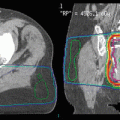
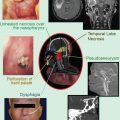
A 55-year-old male previously diagnosed with a Merkel cell carcinoma located on his right side chest wall and receiving 55 Gy in 25 daily fractions using superficial energy photons following excision. The radiotherapy field is well delineated by hypopigmentation, loss of body hair and telangiectasia. Patients manifesting this degree of late radiotherapy cutaneous changes are poor candidates to receive further radiotherapy
4.3 Local Re-irradiation
There is limited preclinical animal data to suggest a retreatment tolerance of >90 % for late dermal necrosis after an 18 Gy single dose and further RT (Simmonds et al. 1989). However, extrapolating this data to the human subject following fractionated cutaneous RT needs to be done with caution. We therefore do not recommend re-local irradiation as a first option but may be considered in select patients.
There is sparse evidence documenting re-irradiation as an alternate option to surgery in the setting of infield (or marginal) recurrence. In a small series of 17 irradiated patients with recurrent HN NMSC (eight BCCs, nine SCCs), all were re-irradiated to a cumulative median surface BED of 103 Gy (range 48.78 Gy–143.5 Gy) and median 108 Gy at 5 mm depth, with 10/17 (59 %) achieving local control and two developing subsequent skin defects (Chao et al. 1995). Median interval from the initial RT was 4 years. The authors concluded that patients receiving an initial BED of <55 Gy at 5 mm depth and a cumulative BED of no >110 Gy at the skin surface had the best outcome, with retreatment fraction sizes limited to 2 Gy to further reduce late tissue reactions. Therefore, the BED of the initial RT and the re-irradiation should not exceed 110 Gy. Interestingly on review of the initial RT details, the authors documented suboptimal initial treatment in 10 of the 17 patients further re-enforcing the need for optimal upfront treatment as a means to reduce the risk of recurrence.
In contrast to the aforementioned series, five lesions were re-irradiated by a Spanish group (no details available) and three recurred with the authors suggesting a benefit in only select patients (Hernandez-Machin et al. 2007).
In a case study of a patient with recurrent auricular SCC previously treated with multiple operations and adjuvant RT, the authors utilized stereotactic body radiotherapy (SBRT) as a means to deliver high-dose (50 Gy in five fractions) re-irradiation and spare surrounding normal tissues. The previous dose fractionation schedule was 60 Gy in 21 fractions. By limiting the treated volume to 2 mm beyond gross tumour, the patient experienced complete clinical regression with no late complications, although the interval from re-irradiation to assessment was not documented. The authors hypothesized that SBRT by delivering large doses per fraction (>10 Gy) may provide a higher likelihood of achieving local control. Of note the 2 Gy BED re-irradiation dose was 100 Gy. This approach is counterintuitive and differs to what many radiation oncologists would recommend when re-irradiating a patient but does indicate that modern techniques of delivering RT such as SBRT and intensity-modulated radiotherapy (IMRT) require further investigation (Brotherston and Poon 2015).
In contrast to re-irradiating using large fractions of RT, 14 patients with secondary-induced angiosarcomas of the breast were re-irradiated utilizing hyperfractionated and accelerated re-irradiation (HART). Multiple abutting electron fields with bolus (post-operatively) or tangential photon fields (preoperatively) were used, and all except one patient received 1 Gy three times daily (4 h interfraction break) with doses varying between 45 and 75 Gy. Previously most patients had received 60 Gy. The authors documented minimal late side effects (four patients with rib fractures) and mild-moderate limb lymphedema with 79 % achieving disease control. In these patients the authors concluded that using a small retreatment fraction of 1 Gy was likely to minimize late normal tissue side effects (Smith et al. 2014).
4.4 Regional Re-irradiation
Regional recurrence after RT, usually adjuvant (i.e. parotid and/or neck metastatic SCC, MCC or MM), poses a more difficult problem, as patients will usually have had a large volume (e.g. ipsilateral parotid ± hemi-neck) of normal tissue (e.g. mandible, soft tissue, brainstem/spinal cord, nerves, carotid artery) irradiated to 50–60 Gy. Following appropriate restaging operable patients should proceed to surgery. For inoperable patients the evidence available for re-irradiation relates predominantly to treating mucosal HNSCC patients. In this analogous setting, recent evidence has emerged supporting the use of highly conformal IMRT (Duprez et al. 2009). Patients retreated with IMRT are likely to have a better outcome (improved locoregional control and decreased severe late effects) compared with conventional 3D conformal re-irradiation. The best results are achieved with radical re-irradiation doses of ~60–70 Gy in 2 Gy daily fractions and retreatment volumes limited to ~2 cm around gross disease or the resection bed. The spine, brainstem and optic chiasm should receive a limited retreatment dose (15–25 Gy) if previously irradiated to tolerance. Of note, even when utilizing IMRT, serious late toxicity and treatment-related deaths are reported in around 20 % of patients. The addition of concurrent chemotherapy to re-irradiation has also been recommended in select patients with mucosal SCC. The role of re-irradiating after salvage nodal surgery is less well defined, but in patients with unfavourable pathology (i.e. close/positive excision margins, extranodal spread), it may be considered.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree