Migraine without aura
A. At least 5 attacks fulfilling criteria B–D
B. Headache attacks lasting 4–72 h (untreated or unsuccessfully treated)
C. Headache has at least two of the following characteristics
a. Unilateral location
b. Pulsating quality
c. Moderate or severe pain intensity
d. Aggravation by or causing avoidance of routine physical activity (e.g., walking or climbing stairs)
D. During headache, at least one of the following
a. Nausea and/or vomiting
b. Photophobia and phonophobia
E. Not attributed to another disorder
Previously used terms: common migraine, hemicrania simplex
Migraine with aura
A. At least 2 attacks fulfilling criteria B–D
B. Aura consisting of at least one of the following, but no motor weakness
a. Fully reversible visual symptoms including positive features (e.g., flickering lights, spots, or lines) and/or negative features (i.e., loss of vision)
b. Fully reversible sensory symptoms including positive features (i.e., pins and needles) and/or negative features (i.e., numbness)
c. Fully reversible dysphasic speech disturbance
C. At least two of the following
a. Homonymous visual symptoms and/or unilateral sensory symptoms
b. At least one aura symptom develops gradually over ≥5 min and/or different aura symptoms occur in succession over ≥5 min
c. Each symptom lasts ≥5 and ≤60 min
D. Headache fulfilling criteria B–D for migraine without aura begins during the aura or follows aura within 60 min
E. Not attributed to another disorder
Previously used terms: Classic or classical migraine, ophthalmic, hemiparesthetic, hemiplegic or aphasic migraine, migraine accompagnée, complicated migraine
Epidemiologic studies described that at least 12 % of the general population suffers from regular migraine attacks [4]. Women are roughly three times more often affected than men. Migraine patients suffer a median of 12 migraine attacks per year; 25 % have at least two attacks per month. Migraine is rated by the World Health Organization among the most disabling chronic disorders [5] and has been estimated to be the most costly neurological disorder in the European countries, largely explained by days missed from work and reduced productivity at work [6]. Acute attack treatments are in less than half of the patients fully satisfactory, and more effective and better-tolerated prophylactic agents to prevent attacks are needed. Mainstream migraine prophylactic treatments have been largely based on serendipitous observations and presumed class effects and have disappointing efficacy and tolerability.
Pathophysiology of Migraine
Understanding of the pathophysiology of migraine starts in acknowledging that migraine is not simply a disease of intermittently occurring pain, but that it relates to processes that over time affect the brain, or act as triggers on a predisposed brain. These processes seem to lead to increased sensitivity or hyperexcitability of brain areas, allowing headache and aura to develop paroxysmally [7]. The mechanisms behind such triggering processes are poorly understood, but are likely multifactorial, and involve several brain structures. Glutamatergic-mediated neuronal hyperexcitability seems an important triggering mechanism [8], but environmental factors (e.g., physiologic factors, stress, weather), comorbid depression, and obesity are also of influence [9].
Mechanisms Behind Migraine Headache
The migraine headache results from activation of the trigeminovascular system (TGVS), which consists of neural connections between dural blood vessels and trigeminal brainstem nuclei [10]. Functional imaging illustrated dynamic involvement of brainstem areas in migraine [11–15]. In the ictal phase, nociceptive (pain) signals are projected from the TGVS to the hypothalamus, the thalamus, and higher CNS centers. These signals are normally controlled by a network involving the hypothalamus and brainstem areas (periaqueductal grey matter and ventral tegmental area) [16, 17]. Paroxysmal dysfunction of (parts of) this network leads to ineffective antinociceptive modulation and vascular control, resulting in the ictal symptoms (pain and autonomic symptoms) [18].
Migraine Aura: Cortical Spreading Depression
The pathophysiological mechanism behind the migraine aura symptoms is cortical spreading depression (CSD). This is a transient depression of activity in neural tissue and is often preceded by a brief burst of action potential firing, which slowly propagates in brain tissue [19–21]. CSD most often involves the occipital lobe, leading to visual symptoms in about 30 % of patients. During the CSD, regional brain hyper- and hypoperfusion (supposed to remain above ischemic thresholds), reductions of blood–brain barrier integrity, and plasma extravasation have been described (see below in section “Neuroimaging Findings in (Prolonged) Aura”). It is unknown why CSD occurs episodically, although temporary changes in excitability seem to be crucial, and likely already develop gradually in hours to days before the symptoms present. The potential role of CSD in activating the migraine headache is compelling in animal experiments but still controversial in humans [22].
Role of Neuroimaging in Understanding the Pathophysiology of Migraine
Effects on the brain (structure, neurochemistry, function) and neurovascular system have been widely documented during the different phases of migraine, and neuroimaging has played a significant role in the current understanding of pathophysiological processes behind migraine. However, these processes are still only partially understood, are likely multifactorial, and involve several brain structures.
Neuroimaging has contributed significantly to the current neuroscientific knowledge on structural and functional brain changes during the ictal phase (aura and headache) of migraine. It is however largely unknown which parts of the brain structurally, functionally, or biochemically change earlier on, during the premonitory phase. The thalamus, hypothalamus, and probably other deep brain and brainstem structures seem to play a role. Further research focusing on such early changes in the premonitory phase may provide insight in when and why brainstem nuclei and pain networks become paroxysmally dysfunctional, how the trigeminovascular system becomes activated, and what pathophysiological changes precede and characterize the aura symptoms. Given the diagnostic focus of this chapter, the neuroscientific neuroimaging findings (based on, e.g., voxel-based morphometry, DTI, MRS, etc.) behind parts of migraine pathophysiology are considered out of scope here and will therefore not be further discussed.
Diagnostic Neuroimaging Indications in Migraine
The diagnosis of primary headaches is exclusively a clinical task based on history taking and careful neurologic examination. In cases of acute headache (like primary thunderclap headache or after trauma, etc.) or suspected symptomatic headache, the need for neuroimaging is mostly evident and will not further be discussed in this chapter. For non–acute headache, as applies to most migraine patients, neuroimaging is overused and is only in selected cases considered to be appropriate.
CT scans and notably MRI scans are frequently requested and performed in migraine patients who seek medical help. Often this is driven by patient’s anxiety about having an underlying pathologic condition, or to improve patient overall satisfaction and medical care. The fact that radiological examinations are not particularly invasive or uncomfortable reduces thresholds further.
However, notably, in patients presenting with typical primary headaches, the very low likelihood of detecting explanatory underlying diseases that change treatment or diagnosis must be considered. Combined results of imaging studies (CT and MRI) in over 3,700 headache patients (not exclusively “typical migraine”) together show a low yield of about 0.4 % in migraine patients [23]. In “typical migraine” patients, the yield is likely to be even lower.
Potential risk of “unnecessary imaging” is the discovery of an incidental finding, which eventually needs further diagnostic workup of follow-up. Further, potential problems include false-positive studies, false reassurance from an inadequate study, allergic reaction to contrast agent, etc. Furthermore, the current resource-restricted medical environment more and more requires evidence-based justification of diagnostic imaging.
AAN Recommendations
In 2000, the quality standards subcommittee of the American Academy of Neurology (AAN) published an evidence-based guideline on the role of neuroimaging in patients with headache [24], to assist the physicians in making appropriate choices in diagnostic workup. A total of 28 studies (from 1966 to 1998) were reviewed. Based on reported rates of “abnormalities related to headache that may require further action” (like acute cerebral infarct, neoplastic disease, hydrocephalus, aneurysm, or arteriovenous malformation) combined with data from patient history and neurological examination, the following symptoms were identified to significantly increase the odds of finding a significant abnormality on neuroimaging in patients with non-acute headache [24]:
Rapidly increasing headache frequency
History of lack of coordination
History of localized neurologic signs or a history such as subjective numbness or tingling
History of headache causing awakening from sleep (although this can occur with migraine and cluster headache)
Based on these findings, the following AAN recommendations for non-acute headache were formulated:
Consider neuroimaging in patients with an unexplained abnormal finding on the neurologic examination (Grade B).
Consider neuroimaging in patients with atypical headache features or headaches that do not fulfill the strict definition of migraine or other primary headache disorders (or have some additional risk factor, such as immune deficiency), when a lower threshold for neuroimaging may be applied (Grade C).
Neuroimaging is not usually warranted in patients with migraine and a normal neurologic examination (Grade B).
EFNS Guidelines
The European Federation of Neurological Societies (EFNS) published a similar guideline for the management of non-acute headache (revision in 2010) [25], also mainly based on review of published evidence. This EFNS guideline includes the following summarized statements relevant for migraine (grade B recommendations):
In adult and pediatric patients with migraine, with no recent change in pattern, no history of seizures, and no other focal neurological signs or symptoms, the routine use of neuroimaging is not warranted.
Exception of these rules should be made in the diagnosis of trigeminal autonomic headaches and headaches that are aggravated by exertion or a Valsalva-like maneuver [23].
In patients with atypical headache patterns, a history of seizures, or neurological signs or symptoms or symptomatic illness such as tumors, acquired immunodeficiency syndrome (AIDS), and neurofibromatosis, MRI may be indicated.
When neuroimaging is warranted, the most sensitive method should be used, and MRI (and not CT) is recommended in these cases.
With a normal unenhanced MRI, in the absence of other diseases and suspicion on metastasis/vasculitis/etc., there is no need for additional scanning with gadolinium.
There is no role for conventional Röentgen techniques.
Digital subtraction angiography is not appropriate in the screening of patients with headache for intracranial disease.
Summarized Recommendations
In the recent meta-analysis by Detsky et al., data from over 3,700 patients were included [23]. The recommendations from this meta-analysis are consistent with the guidelines by AAN and EFNS, although additional recommendations were included to perform imaging in cases (1) with nonvisual of aura (sensory or motor), (2) an aura that has changed in character, or (3) an aura that cannot be clearly described as typical of migraine aura.
Since a 2007 case series demonstrated that even cluster headache with a typical time pattern and an excellent response to typical treatment can still be caused by underlying structural pathology such as a pituitary tumor, patients with trigeminal autonomic headaches should be considered to undergo neuroimaging [26].
In summary, patient history, details of symptoms, and careful clinical neurological examination are together the most important tools in diagnosing and treating migraine. In most patients with non-acute headache, this will lead to a reliable diagnosis (applying the ICHD criteria) and do not require any further laboratory tests or neuroimaging.
Table 2 summarizes the combined recommendations on the use of neuroimaging in non-acute headache patients.
Table 2
Recommendation on the use of diagnostic neuroimaging in non-acute headache
A. Consider neuroimaging in non–acute headache patients with “red flags” |
Unexplained abnormal findings on neurologic examination |
(Atypical) headaches that do not fulfill ICHD-II criteria for primary headaches |
Additional risk factors (e.g., immune deficiency, tumors, etc.) |
History of (associated) seizures |
Recent changes in headache pattern |
Nonvisual (e.g., sensory or motor) or atypical aura pattern |
B. Neuroimaging is not usually warranted in patients with typical migraine with or without aura and normal neurological examination |
C. When neuroimaging is warranted, MRI is method of primary choice |
Migraine and Stroke
Accumulating evidence from the last 3–4 decades has expanded the spectrum of neurovascular pathology linked to migraine. Initial case reports of “migrainous stroke” were followed by retrospective and prospective mostly hospital-based case-control studies assessing the prevalence of clinical ischemic and hemorrhagic stroke in migraine patients, showing a consistent association between migraine with aura and stroke; the association with migraine without aura is less evident [27, 28]. MRI studies have further identified that migraine is also associated with markers of small vessel disease, including (progressive) white matter lesions (WMLs), brainstem T2 hyperintensities, posterior circulation subclinical infarcts, and microbleeds [29–31]. And finally, reports from epidemiological studies on associations between migraine and coronary events [32, 33] and all-cause mortality [34] further illustrate the broad spectrum of lesions associated with migraine, likely to be explained via complex relationships [35].
The relationship between migraine and stroke is complex, and the following paragraphs will expand on different aspects of this relationship; neuroimaging examples will illustrate how migraine patients with (suspicion of) hemorrhagic or ischemic stroke may present.
First, migrainous infarction and its imaging appearances will be described, to be followed by a paragraph on “aura-related” neuroimaging findings, because the clinical symptoms of either “infarction” or “aura” are often very similar in the acute and subacute moments of presentation.
Migrainous Infarction
Kurth et al. suggested that probably the first report on migrainous infarction came from Féré, who described a patient with migraine who died after 2 months of headache, visual disturbances, and hemiplegia [35, 36]. Various case reports of migrainous infarction have been published since then and have made clear that migraine can act as a direct cause of ischemic stroke. In such cases, stroke is assumed to be directly and causally related to an acute migraine attack.
Because it is often impossible by clinical examination alone to differentiate between TIA, prolonged aura, and migrainous infarction, MRI (notably with diffusion weighting) today plays a key role in the diagnosis of migrainous infarction. The current ICHD-II criteria strictly define migrainous infarction as ischemic stroke that occurs when during a typical migraine with aura attack, one or more migrainous aura symptoms persist longer than 60 min, with neuroimaging proof of an associated ischemic brain lesion in an appropriate region and absence of other underlying causes (Table 3).
A. The present attack in a patient with migraine with aura is typical of previous attacks except that one or more aura symptoms persist for >60 min |
B. Neuroimaging demonstrates ischemic infarction in a relevant area |
C. Not attributed to another disorder |
From this definition, it is indirectly evident that migraine patients who present with “prolonged aura symptoms” require an appropriate neuroimaging workup with MRI and, when an acute ischemic lesion is present, additional diagnostic workup to exclude other underlying disorders. In cases of coexisting other causes (like cardiac arrhythmia, coagulation disorders, embolism through a patent foramen ovale, cervical artery dissection), the diagnosis then has to be (changed to) ischemic stroke coexisting with migraine. The same applies when in a patient with a history of migraine without aura, an ischemic lesion develops during or after a migraine attack. In other cases, when criteria are not completely fulfilled, ischemic stroke in a migraine patient may be categorized as cerebral infarction of other causes presenting with symptoms resembling migraine with aura [3].
In the past decades, the diagnostic criteria for migrainous infarction have been changed (ICHD-I vs. ICHD-II), and studies inconsistently applied the criteria. This explains probably the relatively wide range (0.8–3.4 per 100,000) of reported annual incidences and points at a probable amount of overdiagnosis [37]. Although this implies that migrainous infarction is a rare condition, which is further illustrated by Wolf et al. who estimated that it accounts for approximately two among 1,000 “overall” strokes per year [38], it needs to be considered that migrainous infarction predominantly affects younger patients. In that age category, migrainous infarction was estimated to account for 13 % of first-ever ischemic strokes [39].
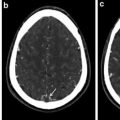
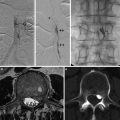
Neuroimaging Findings in Migrainous Infarction
The largest series of migrainous infarction cases to date are reported by Wolf et al. (17 cases) [38] and Laurell et al. (33 cases) [40]. In both reports, patients underwent an appropriate stroke workup, and diagnoses according to ICHD-II criteria were reported. Table 4 shows the main study findings.
Table 4
Recent case series of migrainous infarction
Laurell et al. [40] | Wolf et al. [38] | |
|---|---|---|
Cases | n = 33 (all ICHD-II) | n = 17 (n = 11 ICHD-II)a |
M:F | 39 %:61 % | 23 %:77 % |
Age at stroke onset | 19–76, median 39 years | 20–71, mean 45 years |
Posterior circulation | 82 % | 71 % |
Cerebellum | 21 % | 6 % |
Multiple lesions | 41 % | |
Family history of migraine | 75 % | 24 % |
Patent foramen ovale | 40 % | 65 % |
Both studies reported a clear predominance of infarcts in the posterior circulation, supporting previous observations. The low age at stroke onset is a further key finding in both studies; therefore, when treating a young patient presenting with stroke, migrainous infarction should be kept in mind. In both studies, outcome was relatively favorable. Although no studies have systematically examined the appearance of migrainous infarcts, notably when supratentorially, from a number of case reports, it is suggested that the ischemic insults predominantly may affect the cortex. Similarly, cortical ischemia that crosses different vascular territories may also point at a migrainous infarct mechanism, but this is probably an infrequent finding, since in the study by Wolf et al., no such “crossing” lesions were identified, neither on DWI nor on PWI [38]. However, aura-related hypoperfusion (see below) quite typically seems to cross territories.
The underlying mechanisms of migrainous infarction are unknown, but are probably related to CSD-related changes, including hypoperfusion, and changes in blood–brain barrier permeability (which might lead to an exacerbation of local cellular injury caused by ischemia). Together with factors predisposing to coagulopathy and release of vasoactive neuropeptides, further changes in cerebral hemodynamics, arterial thrombosis, and infarctions may be explained [41].
In the Figs. 1, 2, 3, and 4, case descriptions illustrate various presentations and appearances of migrainous infarcts. In Fig. 5, a case with “cerebral infarction presenting with symptoms resembling migraine with aura” is described and illustrates that it can be difficult to apply a correct and meaningful diagnosis, given the strict ICHD-II criteria.
Neuroimaging Findings in (Prolonged) Aura
According to the ICHD-II criteria, a diagnosis of persistent aura without infarct can be applied when aura symptoms remain present longer than 1 week and when there is no neuroradiological evidence of ischemia. This is a rare condition that seems to affect genetic forms of migraine (like familial hemiplegic migraine) somewhat more often.
In “regular” migraine with aura patients, aura symptoms incidentally also persist longer than 60 min. When more than one aura symptom is present (e.g., visual and sensory symptoms together or in succession) for each type, 60 min may be accepted. When aura persists longer, this might point at migrainous infarction, although most often symptoms still spontaneously normalize, and probably, only incidentally, patients will be scanned. Imaging studies in such “non-infarct” cases by definition do not show ischemic changes, but various case reports and series have described other CSD- or aura-related effects on the brain tissue and neurovascular system that will be discussed in the next paragraphs.
Cutrer et al. [42] and Sanchez del Rio et al. [43] studied spontaneous migraine episodes with perfusion-weighted MRI, including six patients studied during regular (not prolonged) visual aura, within 31 ± 6 min after the onset of visual symptoms. In all studies, perfusion deficits were observed in the occipital visual cortex from which the hemifield defect was originating. Maximum measured changes were 37 % decrease in CBF, 33 % decrease in rCBV, and 82 % increase in MTT. Several small series and case reports have been published since then, mostly with consistent findings of mild regional hypoperfusion, uni- or bilaterally affecting overlapping vascular territories.
Förster et al. reported a prospective study in which patients with suspected acute ischemic stroke were evaluated. In this study, 33 patients with a final diagnosis of migraine with aura were compared with age-matched patients with a final diagnosis of acute ischemic stroke [44]. As a consequence of the study methodology, in this cohort, the number of patients with “rare” aura symptoms (like hemihypesthesia, hemiparesis, aphasia) was overrepresented, as well as “acute onset” of symptoms. In 54 % (n = 18) of migraine with aura patients, PWI showed hypoperfusion that was involving more than the PCA territory in all but one of the patients, although the PCA territory was predominantly involved in 61 % of cases. In seven patients (39 %), the hypoperfusion extended to the parietal, temporal, or frontal lobe. There was no clear association between clinical symptoms and location of perfusion changes. There were no diffusion-weighted imaging (DWI) abnormalities related to the aura symptoms, and there were no vessel occlusions or stenoses on MRA. In comparison with acute ischemic stroke patients, the aura patients more often had hypoperfusion involving more than one territory and less increased TTP and MTT ratios.
A few reports pointed at the occurrence of “crossed-cerebellar diaschisis” in cases with aura-related perfusion changes. Dodick et al. reported a migraine patient with typical attacks of sensory aura during 30–60 min followed by headache, who showed reversible reduction in CBF in the left cerebral hemisphere and associated crossed-cerebellar diaschisis (hypoperfusion in the right cerebellar hemisphere) during a typical attack on a brain SPECT scan [45]. Iizuka et al. observed a case with prolonged aura crossed-cerebellar hyperperfusion on brain SPECT scan on day 2 after symptom onset, which was explained by the authors as a consequence of uncoupled hyperperfusion with low function in the left cerebral hemisphere (corresponding to the neurological deficits). Both the crossed hyperperfusion and hypoperfusion illustrate the possibility of associated flow (and metabolic) alterations distant to and opposite of the primary site of disturbances, which could be relevant in understanding the occurrence of silent ischemic lesions in the cerebellum in migraine patients (see later).
Besides flow alterations, a variety of other imaging findings have been described on brain imaging during (prolonged) migraine aura that all together may be explained by temporary changes in blood–brain barrier function that are probably secondary to aura-related cortical spreading depression and/or spreading hypoperfusion. A number of reports described “vasogenic leakage,” that may present as regional sulcal hyperintensity on native FLAIR MRI images [46], as (delayed) subarachnoid (sulcal), leptomeningeal, or cortical gadolinium enhancement on FLAIR or T1-weighted MRI scans [47–50] or as increased vascular permeability on perfusion-weighted MRI [51]. In few cases with (prolonged) aura, symptoms may be associated with reversible cortical swelling and hyperintense signal changes on FLAIR images, distributed along the regional cortical ribbon corresponding to the symptoms, without clear diffusion restriction (vasogenic edema) [52].
Migraine as a Risk Factor for Clinical Ischemic Stroke
Besides the (rare) presentation of “migrainous infarction” (i.e., directly related to a migraine attack), migraine patients have also a higher chance to present with an ischemic or hemorrhagic stroke unrelated to a migraine attack.
Over the past four decades, many observational studies, hospital-based stroke case-control studies, and population-based studies have evaluated the association between migraine and clinical ischemic stroke. Meta-analyses [27, 53] of these studies summarized that migraine patients are about at doubled increased risk, which notably applies to patients with migraine with aura. Schürks calculated a pooled relative risk of 1.7 (95 % CI 1.3–2.3) for migraineurs compared to controls [27]. From eight studies, data specified by migraine subtype was available, resulting in a pooled relative risk of 2.2 (95 % CI: 1.5–3.0) for patients with migraine with aura and 1.2 (95 % CI: 0.9–1.7) for patients with migraine without aura, compared to controls. A higher migraine frequency seems also to further increase the risk of ischemic stroke. Due to the lower prevalence of migraine in men, the association between migraine and ischemic stroke is less certain in men.
The risk is highest in women, notably at younger age (<45 years), and in this group, the risk further increases (up to 10x increased risk) with concurrent use of oral contraceptives. In individual studies, effects of concurrent hypertension and heavy smoking showed a greater than multiplicative effect on the risk [54, 55]. Associations with markers of endothelial dysfunction and factors linked to prothrombotic or proinflammatory conditions have also been suggested as explanatory for the increased stroke risk in migraine [56]. Genetic factors, including polymorphisms in the MTHFR, ACE, MEPE, and IRX4 genes, have been linked with both migraine and ischemic stroke, but further studies need to assess the pathophysiological relevance of such findings.
Topographic and Pathophysiological Considerations
Few structured data exist on the topography of non-migrainous infarcts in migraine patients. In a large series of 3,500 patients with acute stroke, 130 (3.7 %) had active migraine, and about 50 % of these were <45 years of age. In the younger patients, posterior circulation involvement (55 %) was characteristic [57]. From case reports and small series, it was also suggested that the occipital lobe and/or the posterior cerebral artery territory seem to be overrepresented in migraine patients with clinical ischemic stroke. Recently, Oygarden et al. retrospectively investigated whether the lesion pattern on diffusion-weighted MRI scans was different between migraineurs (with and without aura; n = 196) and non-migraineurs (n = 720) with clinical ischemic stroke [58]. In the migraine group, younger patients and women were overrepresented, and infarcts were more often due to cardioembolism and less often due to small vessel disease than in the control group. Migraine patients presented more often with symptoms from the posterior circulation and had more cortical (OR 1.8 CI: 1.3–2.5), small (OR 1.9 CI: 1.04–3.5), cerebellar (P = 0.026), and occipital infarcts (13.3 % vs. 8.6 %; P = 0.05). Migraine patients with infarcts had a three times higher chance to have a patent foramen ovale (PFO).
Oygarden et al. suggested that the higher frequency of smaller lesions in migraineurs with aura may point at a higher vulnerability to damage following minor ischemic insults (similarly as was described in experiments in mice with familial hemiplegic migraine) or that ischemia in migraineurs may simultaneously lead to a small “clinically silent” ischemic lesion (that normally would not be noticed) and ischemia-triggered CSD resulting in “clinical” stroke like neurological deficits leading to hospital admission [58].
Although the earlier study results were criticized (e.g., because of potential misclassification, referral bias, lack of neuroimaging proof, etc.), the consistent findings in several studies over the years have migraine set today as an acknowledged ischemic stroke risk factor. It has to be noted that in absolute terms, stroke in young women with migraine (estimated at 5.5/100,000 annually) [59] remains rare, although at the same time, in this age group, migraine should be considered as an important risk factor.
Migraine as a Risk Factor for Clinical Hemorrhagic Stroke
Data on a possible association between migraine and hemorrhagic stroke have remained inconsistent for years. However, in 2013, Sacco et al. meta-analyzed a total of four case-control and four cohort studies that together included 320,539 individuals in whom 1,600 intracerebral or subarachnoid hemorrhages occurred [28]. Migraine patients were found to be at significantly increased risk for hemorrhagic stroke (OR 1.48; 95 % CI 1.2–1.9; P = 0.001). There was no clear difference between migraine with aura (OR 1.6; 95 % CI 0.9–3.0; P = 0.13) or migraine without aura (OR 1.4; 95 % CI 0.7–2.62 P = 0.3), male and female patients, or younger and older subjects with migraine, although Kuo et al. reported higher risks of migraine with aura in male patients and patients <45 years of age [60]. A limitation in the review by Sacco et al. was that subarachnoid and intraparenchymatous hemorrhages could not be separated. Next to ischemic stroke, migraine patients are thus likely also at increased risk for hemorrhagic stroke, although mechanisms likely differ and remain to be elucidated, and subgroups most at risk still need to be identified.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree