Fig. 1
General study conduct of PRRT at the department of nuclear medicine, medical university of innsbruck. Within the music project different types of music were applied via MP3 players during the time of hospitalisation, starting the day before application of PRRT. STAI questionnaires were used before and after infusion of radioactivity on the same day, BSI forms were reported on the day of admission as well as on the day of remission from the hospital. Patients of the control group underwent the questionnaires at the same time points
Since SST receptor scintigraphy has been introduced into the clinical routine various SST analogues were developed with differences in the biodistribution and radiation doses to the organs. 111In-DTPA-OC as well as of 111In-/90Y-DOTA-TOC differs from that of 111In-/90Y-DOTA-LAN in terms of higher liver and kidney, and less bone marrow uptake (Virgolini et al. 2009). Evaluation of the type of radiotracer to be used for PRRT should be based on the scintigraphic pattern and on dosimetric data which also provide quantitative values for confirmation of visual evaluation.
The individualised treatment regime applied at our department is also based on various patient related factors. In patients with advanced disease stages, older age, poor Karnovsky performance index and diminished organ function due to pre-treatment with chemotherapy individual doses are adapted guided by individual parameters, so that severe intermittent side effects of PRRT may be avoided. Patient receives an amino acid solution for kidney protection (Bodei et al. 2003). The amino acid solution is applicated over 6 h starting 30 min before administration of the radiopharmaceutical via a separate line.
2.2.2 Inclusion Criteria and Evaluation of Therapy Outcome
For initial evaluation, 68Ga-DOTA-TOC PET was performed for accurate staging and evaluation of the SSTR status of the lesions (Gabriel et al. 2007, 2009).
If PET is positive, either with 68Ga-DOTA-TOC or 68Ga-DOTA-LAN (in case of a negative or insufficient 68Ga-DOTA-TOC binding) and visual scoring revealed higher uptake in the lesions as compared to the normal liver uptake, patients are included for PRRT. Dosimetry using the 111In-labelled compound is followed to calculate tumour and organ doses.
Normally, the 90Y-labelled compound is used as the first treatment choice. However, if individual lesions are smaller than 2 cm in diameter 177Lu-DOTA-TATE is preferred for PRRT. This compound is also used in patients who have already undergone PRRT with 90Y-DOTA-TOC and progression as second choice tracer due to lower radiation dose to the kidneys (Forrer et al. 2005).
The reference standard for the clinical outcome after PRRT is based on follow-up controls with all available patient records including laboratory parameters and the correlation of PET and CT/MRI findings according to Response Evaluation Criteria in Solid Tumours(RECIST) (Öberg and Jelic 2008; Duffaud and Therasse 2000). If necessary, other imaging modalities such as lesion guided MRI, and 18F-fluorodesoxyglucose (18F-FDG) PET, endoscopy and ultrasonography are also used to assess response to PRRT (Gabriel et al. 2009; Putzer et al. 2009).
Findings are summarized to three categories: response to therapy, stable disease (SD) and progressive disease (PD). Complete response, minor (MR), i.e. tumour shrinkage of less than 30 %, and partial response (PR).
Adverse effects of PRRT (evidence for thrombocytopenia, leukopenia, haemoglobinaemia, anaemia and elevation of creatinine) during the therapy course are reported.
2.2.3 Results: Experience Innsbruck
2.2.3.1 Efficacy
Overall, 33 % (33/100) of patients had documented remission, including 10 patients with MR and 23 patients with PR. Forty-two patients (42 %) showed SD (overall response rate 75 %) (Fig. 2). Results related to the used radiopharmaceutical are depicted in Table 1.
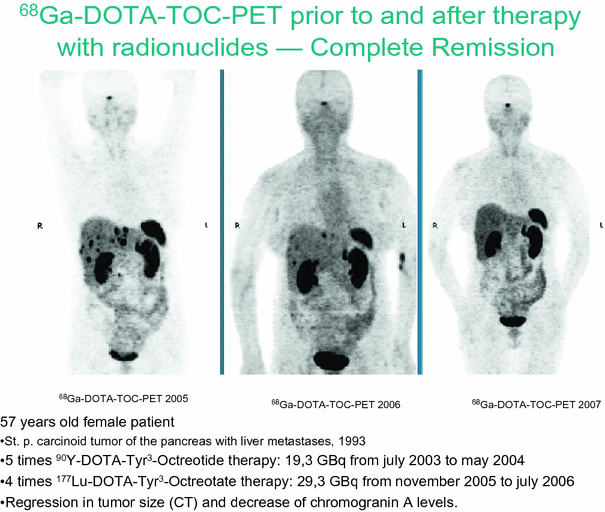

Fig. 2
Figure 1 shows the 68Ga-DOTA-TOC PET scans of a 57 year old female patient suffering from a carcinoid tumor of the pancreas with liver metastases, which was diagnosed in 1993. She was referred to our institution in 2005 because of recurrence of the disease, including liver metastases. 68Ga- DOTA-TOC PET in 2005 (see image on the left) showed intense somatostatin receptor expression regarding the liver metastases. The patient then received 5 times 90Y-DOTA-Tyr3-Octreotide therapy with a cumulative dose of 19.3 GBq from july 2003 to may 2004. The patient represented at our institution in 2006 for a follow-up visit, and 68Ga-DOTA-TOC PET (image at the center) in 2006 showed a partial remission. However, some liver metastases still showed high SSTR expression. Therefore, the patient received 4 applications of 177Lu-DOTA-Tyr3-Octreotate therapy with a cumulative dose of 29.3 GBq from november 2005 to july 2006. The 68Ga-DOTA-TOC PET follow-up in 2007 (image on the right) resulted in a full remission in the PET and a decrease in tumor size (CT) and decrease of chromogranin A levels
Table 1
Results of PRRT-innsbruck experience
MR | CR/PR | SD | Response (%) | PD | |
|---|---|---|---|---|---|
90Y-DOTA-TOC (n = 60) | 3 | 12 | 32 | 47/60 (78) | 13 |
177Lu-DOTA-TATE (n = 55)a | 8 | 15 | 19 | 42/55 (93) | 13 |
2.2.3.2 Toxicity
In 32 patients no adverse effects after the administration of the radiopharmaceuticals were observed. In the other 68 patients various delayed toxicities occurred. 56 % showed side effects WHO grade 1. Moderate side effects—WHO grade 2—were observed in 6 % of patients. Both groups did not require further supportive treatment and recovered spontaneously. However, 6 patients needed further therapeutic intervention because of their severe haematological side effects including four patients with WHO grade 3 and 2 patients with grade 4 toxicity.
Acute adverse effects during 24 h after the administration of the radiopharmaceutical were observed in 37 patients with abdominal discomfort and/or vomiting. These side effects are considered to be related to the amino acid solution for kidney protection.
In 16 patients with diarrhoea due to functional NET and elevated tumour markers (chromogranin A) the frequency and severity of diarrhoea markedly increased up to a period of 10 days post therapeutically possibly related to hormonal release.
Severe long-term nephrotoxicity was observed in none of the patients. In only 12 patients elevation of the creatinine levels were observed and no patient suffered from treatment-related kidney failure.
2.2.4 Conclusion
In recent years, different therapeutic approaches have emerged for individualised targeted therapy in tumour patients. The expression of SST receptors on NETs has proven useful for imaging of NETs in different clinical situations, e.g. for initial diagnosis, staging and follow-up, with high diagnostic efficacy. Based on these findings tumours were also treated by SST-based radiopharmaceuticals showing promising overall results.
In our department patients are selected for PRRT by their inherent biological characteristic which can be evaluated by scintigraphy using either DOTA-TOC or, if this scan is negative, with DOTA-LAN. Recently, these compounds have also been labelled with gallium-68 for PET imaging providing higher diagnostic quality for initial evaluation as well as for the follow-up scanning after PRRT (Gabriel et al. 2007, 2009). Individualised therapy in nuclear medicine especially requires optimal functional imaging techniques with PET for visual evaluation and scintigraphy with 111In labelled compounds for dosimetry.
PRRT can be considered in patients with inoperable metastatic disease and a positive SSTR scan. Although several studies have shown promising overall results, reported anti-tumour effects vary considerably. The individualised treatment regime applied at our department is based on various patient-related factors applying sometimes reduced standard doses. Nevertheless, our approach shows response to therapy in 75 % of patients which is basically comparable to published data of other centres (for review see Virgolini et al. 2009, van Essen et al. 2009) In addition, half of our patient population showed SD, and even in the patients with PD under therapy some palliation of tumour-related symptoms can be observed. In patients with advanced disease stages, older age, poor Karnovsky performance index and diminished organ function due to pre-treatment with chemotherapy, treatment doses are adapted according to clinical and laboratory parameters, so that severe, mostly intermittent, side effects (WHO grade 3 and 4) of PRRT are only rarely seen.
An other explanation for rather good tolerance of this regime might be based on the combination of PRRT with the cold SST analogue which could be a protective factor for kidney function. Furthermore, in about 30 % of patients a further improvement of the objective therapy response was also observed up to 12 months after finalization of PRRT. This might be in part contributed by the continuous application of the cold analogue to make the application of the non-labelled SST analogue an integral component of our regimen.
3 The Neuroendocrine Tumour Patient and “Restitutio ad Integrum”
“Restitutio ad integrum” (lat. integritas) means “full healing” or “complete recovery”. NET Patients undergoing PRRT are mostly heavily metastasised and put a lot of hope into this new therapy modality. However, the treatment with high dose radioactivity is not so common and may cause a certain anxiety level. There is a lot of misinformation around and only a few studies exist in the area of external beam radiation therapy linking radiation to the patient general well-being or anxiety level (Smith et al. 2001). For nuclear medicine therapy, to our knowledge, there exist even less published data in this area. In NET patients undergoing PRRT, some authors (Teunissen et al. 2004) have reported significantly improved quality of life using the questionnaire from the European Organisation of Research and Therapy in Cancer Quality of Life Questionnaire C30, EORTC QLQ-C30 (Therasse et al. 2000). This questionnaire covers the global well-being of the patient. Other centres have also reported similar improvement of quality of life parameters in NET patients. In general, our view gets more and more accepted that for a NET patient a good clinical condition with stable tumour disease is better than a reduction in tumour size with reduced quality of life by high dose PRRT.
Healing, assessed physically, is the process by which the cells in the body regenerate and repair to reduce the size of a damaged or necrotic area. Healing incorporates both the removal of necrotic tissue, and the replacement of this tissue. Healing, assessed spiritually, emotionally, mentally or otherwise, is a process which involves more than just the action of cells (http://psychology.wikia.com/wiki/Healing).
When building the therapy unit at the Department of Nuclear Medicine at the University of Innsbruck we aimed to establish a division in which the patient is treated not only by high standard nuclear medicine care but we also wanted to integrate other aspects of patient care.
The principle of architecture and healing (http://nuklearmedizin.i-med.ac.at/, www.youtube.com/watch?v=D15LOMR-aJY) was adapted to our therapy unit. The emphasis of healing, as mentioned above, was based on John 8,12 “I am the light of the world…. He, who follows Me, shall not walk in darkness, but have the light of life” as well as on Wolfgang Goethe` s last words “light, more light”. Consequently, the logo of our department (Fig. 3) depicts a severe sick patient who is in dark and who becomes enveloped by healing light, so that he can be transformed into well-being strength and joy. The various wall colours of our rooms where our NET patients are treated by PRRT (Fig. 4) are in perfect harmony with many Tyrolean Swarowski stones placed on the walls.



Fig. 3
Logo of the department of nuclear medicine, medical university of Innsbruck

Fig. 4
The principle of architecture and healing is present in all aspects of our therapy ward. The various wall colours of our rooms for PRRT resonate with many Swarovski stones, which are part of the decoration in each treatment room
The ubiquity of music in human culture is indicative of its ability to produce pleasure and reward value. Recent PET studies have indicated that activity in paralimbic brain regions correlate with unpleasant or mildly pleasant emotions elicited by varying amounts of musical dissonance (Blood and Zatorre 2001), while subject-selected music that “chills” is accompanied by changes in heart rate, electromyogram and respiration and is correlated to brain regions thought to be involved in reward/motivation, emotion, and arousal (such as ventral striatum, midbrain, amygdala, orbitofrontal cortex, ventral medial prefrontal cortex). This brain structures are known to be active in response to other euphoria-inducing stimuli such as food, sex, etc., and link music with biologically relevant stimuli via their common recruitment of brain circuitry involved in pleasure and reward.
Anxiety is an understandable response to a diagnosis of cancer and its treatment. Two distinct types of anxiety are often presented, either acute or chronic. Roughly, half the cancer patients present with anxiety at varying levels and anxiety is simply underestimated. The stage of the cancer is a particularly important factor influencing anxiety. Specific music selections or types of music may have different effects for different people, and may have different effects for the same person at different times. Self-administered music interventions allow patients freedom to schedule their music intervention at times when it is most convenient (see Sect. 4) (Siedliecki et al. 2006; Blood et al. 2001)
4 Effects of Music in NET Patients Undergoing PRRT
4.1 Study Aims
We refer to a recently conducted study at the Department of Nuclear Medicine at the Medical University of Innsbruck (Staudinger 2008). In this study, we tested whether “music” can affect the self-reported clinically relevant psychological symptoms and the state anxiety in NET patients undergoing high dose PRRT.
4.2 Study Design
4.2.1 General Study Conduct and Assessment of Outcome Variables
The study was conducted randomised and single-blinded in 37 patients with histologically proven NET. In all patients written informed consent was obtained, and all patients were inwarded for treatment with high dose radiolabeled SST Analogues. PRRT was performed either with 90Y-DOTA-TOC, 177Lu-DOTA-TATE or with 90Y-DOTA-LAN according to our institutional standard criteria (see Sect. 2). The Brief-Symptom-Inventory (BSI) (Derogatis et al. 1983; Franke 2000) as well as the State-Trait Anxiety-Inventory (STAI) (Laux et al. 1981; Spielberger 1972; Spielberger et al. 1977) questionnaire both were used in all patients for assessment of outcome variables.
With the BSI 53 items are reported covering 9 clinical symptom dimensions: somatization, obsession-complusion, interpersonal sensitivity, depression, anxiety, hospitality, phobic anxiety, paranoid ideation and psychoticism, and 3 global indices of distress: (Global Severity Index; Positive-Symptom-Total, and Positive-Symptom-Distress-Index). The global indices measure current or past level of symptomatology, intensity of symptoms and number of reported symptoms, respectively.
With the STAI the acute state anxiety is reported. The STAI differentiates between the temporary condition of “state anxiety” and the more general and long-standing quality of “trait anxiety”. The STAI was therefore used directly before and after the end of infusion of radioactivity.
In Fig. 1, the study conduct is presented. NET patients assigned for PRRT were inwarded, and treatment was applied in various cycles allowing 8–12 weeks between the treatment cycles. Unlabelled octreotide (Sandostatin LAR®) or lanreotide (Somatoline®) was given between the radioactive cycles (Fig. 1a). Each patient received an amino acid solution during PRRT for kidney protection. All patients were infused with 1500 mL mineral solution containing 25 g of l-lysine and 25 g l-arginine. The amino acids solution was applicated over the course of 6 h starting 30 min before infusion of the radiopharmaceutical via a separate line.
Patients of this study received the “music” during one of the PRRT cycles, after the initial assessment of the BSI form (Fig. 1b). The BSI form was repeated after remittance from the therapy unit. The STAI form was used directly before the radiopharmaceutical was applied as well as right after infusion of radioactivity. Patients from the control group (Fig. 1c) underwent the same questionnaires at the same time points.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








