Fig. 1
In order to facilitate LR administrations, a catheter into the SCRC and connected with a subcutaneous reservoir is permanently implanted during surgical procedure
The rationale for intracavitary administered therapies for malignant glioma patients is based on two fundamental factors. First, locally administered therapeutics may circumvent the blood–brain barrier (BBB) and thus potentially achieve higher intratumoural concentrations than are achievable following systemic administration. Second, systemic exposures associated with LR therapies are typically minimal, leading to less systemic toxicity.
Clinical experiences using LR techniques to treat malignant gliomas, including intra-tumour injection of interleukin-2, lymphokine-activated killer cells, toxins and various chemotherapeutic agents, have been reported over the last 30 years.
2.2 Locoregional Radioimmunotherapy in HGG
Theoretically, RIT could be exploited in all those tumours for which a specific MoAbs is available to target its specific antigen. HGG represent the most favourable model because it is refractory to conventional treatments and a suitable marker, the antigen tenascin-C (hereinafter referred to as tenascin), is abundant in the stroma of these neoplasms, but not in normal brain tissue.
Tenascin is a 6-armed glycoprotein that is over expressed in the extracellular matrix of gliomas and other malignancies. Tenascin is located primarily around tumour blood vessels, with this feature becoming more predominant with advancing tumour grade. Several antitenascin moAbs, which bind to different epitopes on the tenascin molecule, have been used for targeted radiotherapy. Antibody BC-2 binds to an epitope found on both the A1 and A4 alternatively spliced fibronectin type III repeats, and mAb BC-4 reacts with an epitope in the epidermal growth factor-like repeat found on all tenascin isoforms. The 81C6 moAb reacts with an epitope present within the alternatively spliced fibronectin type III CD segment (Paganelli 2006).
Radioimmunotherapy, as systemic or locoregional application has the potential to become an option in the management of HGG, complementing the above-mentioned treatment regimens. One of the main therapeutic advantages of radiolabelled moAbs is their potential to overcome the problem of tumour heterogeneity. Because the radionuclides can penetrate up to several millimetres of tissues, radio-emission can kill those antigen-negative tumour cells, which have no specific radiolabelled antibody localised on their surface (cross-fire effect).
Many early RIT trials on brain tumour involved the intravenous administration of radiolabelled moAbs. Although some positive responses were described, more encouraging survival benefits have been reported when the radiolabelled moAbs were administered locoregionally, either into non-resected tumour or into the SCRC.
2.2.1 The Experience of Duke University Medical Center
Investigators at Duke university medical center have assessed the potential therapeutic benefits using the 81C6 moAb labelled with 131I-labelled in patients with GBM and other malignant brain tumours. Preliminary diagnostic-level studies have demonstrated that delivery of radiolabelled moAbs by intravenous route would not yield therapeutically relevant tumour doses without unacceptable toxicity for patients. For these reasons, RIT trials with anti-tenascin moAbs have involved intra-compartmental (locoregional) administration of the labelled protein, into either tumour, spontaneous tumour cysts, or, most frequently, surgically created glioma resection cavities.
In the first phase I study, 42 patients with recurrent glioma were included and the maximal tolerable dose (MTD) was assessed in a dose-escalation study after intracavitary administration of 131I-labelled 81C6. This study showed that the MTD was 100 mCi, with neurotoxicity being the dose-limiting factor. The results of this study suggested that there was a potential survival benefit, as compared to patients treated with stereotactic radiotherapy and high-dose brachytherapy (a median survival of 60 weeks in the present study, as compared to 41 and 46 weeks, respectively). In the second study, 42 patients with newly diagnosed glioma were included in order to investigate dosimetry and dose–response relationships. In these patients, the MTD was 120 mCi, with neurotoxicity being the dose-limiting factor. The median survival of these patients was 79 weeks, as compared to 46 weeks of historic controls, when patients were treated with surgery, chemotherapy and radiotherapy. Based on these encouraging results, a phase II trial was performed in 33 patients with newly diagnosed, previously untreated patients. The median survival after treatment with 120 mCi of 131I-labelled 81C6 in this study was 79–85 weeks, depending on the pathologic type of glioma (patients with astrocytic oligodendroglioma showed a better response than those with glioblastoma multiforme). When 100 mCi of radiolabelled antibody was administered to 43 patients with recurrent glioma, survival was still 69 weeks. The results of these trials warranted a phase III trial, which is currently ongoing.
Subsequently, a human/mouse chimeric mAb, originating from 81C6, was developed, showing better tumour targeting in animal studies. The targeting capabilities of the antibody were subsequently tested in a phase I study that included 47 patients with recurrent disease. This chimeric antibody showed a prolonged retention time within the SCC, as compared to the antibody of murine origin. Owing to the enhanced circulatory half-life of the chimeric antibody, an MTD of 80 mCi was found, as compared to 120 mCi found in previous studies with the murine antibody. In this phase I dose-escalation study, the median survival was 87 weeks for patients with newly diagnosed glioma and 65 weeks for those patients with recurrent disease (Reardon 2006; Zalutsky 2005).
A more recent clinical experience from the same group was concerning the use of alpha-particles instead of beta-particles in LR RIT. Eighteen patients were treated with 211At-labelled chimeric 81C6 (211At-ch81C6) administered into a surgically created resection cavity and then with salvage chemotherapy. Serial gamma–camera imaging and blood sampling over 24 h were performed. A total of 96.7±3.6 % (mean±SD) of 211At decays occurred in the SCRC, and the mean blood-pool percentage injected dose was ≤0.3. No patient experienced dose-limiting toxicity, and the maximum tolerated dose was not identified. Six patients experienced grade 2 neurotoxicity within 6 weeks of 211At-ch81C6 administration; this neurotoxicity resolved fully in all but one patient. No toxicities of grade 3 or higher were attributable to the treatment. No patient required repeat surgery for radionecrosis. The median survival times for all patients, those with glioblastoma multiforme, and those with anaplastic astrocytoma or oligodendroglioma were 54, 52 and 116 weeks, respectively. The authors concluded that the regional administration of 211At-ch81C6 was feasible, safe and associated with a promising antitumour benefit in patients with malignant central nervous system tumours (Zalutsky 2008).
2.2.2 The Experience of European Institute of Oncology, Milan
One of the limitations of directly labelled moAbs for targeted radiotherapy is that as a consequence of their molecular size they diffuse slowly through tissue, hampering their delivery to tumour cells distant from the site of injection. Furthermore, when moAbs are labelled with a high energy isotope, significant radiolysis occurs during the interval between radiolabelling and injection, with consequent decrease in immunoreactivity.
An attractive strategy to compensate for the large size of moAbs is pre-targeting, a procedure in which the moAb is administered first, followed after an appropriate time interval by the injection of a radiolabelled low molecular weight vehicle.
Over the last two decades, nuclear medicine physicians at the European institute of oncology have developed and applied a “3-step” pre-targeting method, both in systemic and locoregional treatment of recurrent gliomas.
The “3-step” method takes advantages of the extremely high affinity between avidin and biotin. Avidin is a small oligomeric protein made up of four identical subunits, each bearing a single binding site for biotin (vitamin H). They can therefore bind up to 4 mol of biotin per mole of protein. The affinity of avidin for biotin is extremely high, with a dissociation constant of the avidin-biotin complex in the order of 10−15 M. For practical purposes, their binding can be regarded as irreversible. Briefly, the 3-step-based RIT is based on the sequential administration of a specific biotinyled antibody, avidin an radioactive biotin.
2.2.2.1 3-Step Locoregional Radioimmunotherapy
After the encouraging results from the early experiences in gliomas, using the intravenous route (Paganelli 1999; Grana 2002), investigators at the European institute of oncology (Paganelli 2001) experimented the 3-step pre-targeting method also for locoregional applications.
In a phase I–II study the safety profile and antitumuor efficacy of the 3-step method in the locoregional therapy of recurrent high grade gliomas was assessed. Twenty-four patients with recurrent HGG (8 AA and 16 GBM) underwent second surgical debulking with implantation of an indwelling catheter (connected with a subcutaneous reservoir) into the SCRC, in order to receive the radioimmunotherapeutic agents.
Biotinylated anti-tenascin moAbs (BC2 or BC4), avidin and, finally, 90Y-Biotin were subsequently injected through the catheter. Each patient received two of these treatments 8–10 weeks apart and the injected activity ranged from 0.5 to 1.1 GBq. Dosage was escalated by 0.2 GBq in four consecutive groups. Bremsstrahlung images were acquired to confirm the correct localisation of the 90Y-biotin (Fig. 2). The treatment was well tolerated without acute side effects up to 0.7 GBq. The maximum tolerated activity was 1.1 GBq limited by neurologic toxicity. None of the patients developed haematological toxicity. In three patients infection occurred around the catheter. The average absorbed dose to the normal brain was minimal compared with that received at the surgical resection cavity interface.
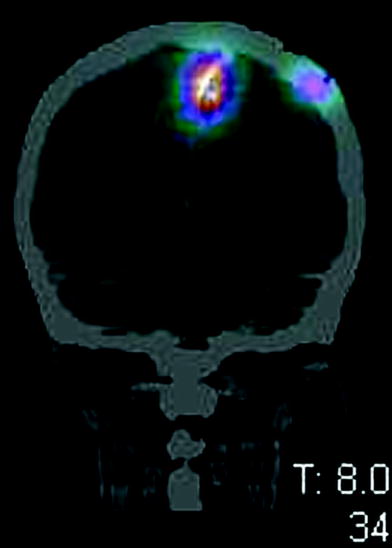

Fig. 2
Bremsstrahlung brain SPECT-CT confirms the correct localisation of the 90Y-biotin locoregionally injected into the SCRC
This study assessed that with activity ranging from 0.7 to 0.9 GBq per cycle, “3-step” LR RIT was safe and produced an objective response (partial and stable disease in 75 % of patients).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







